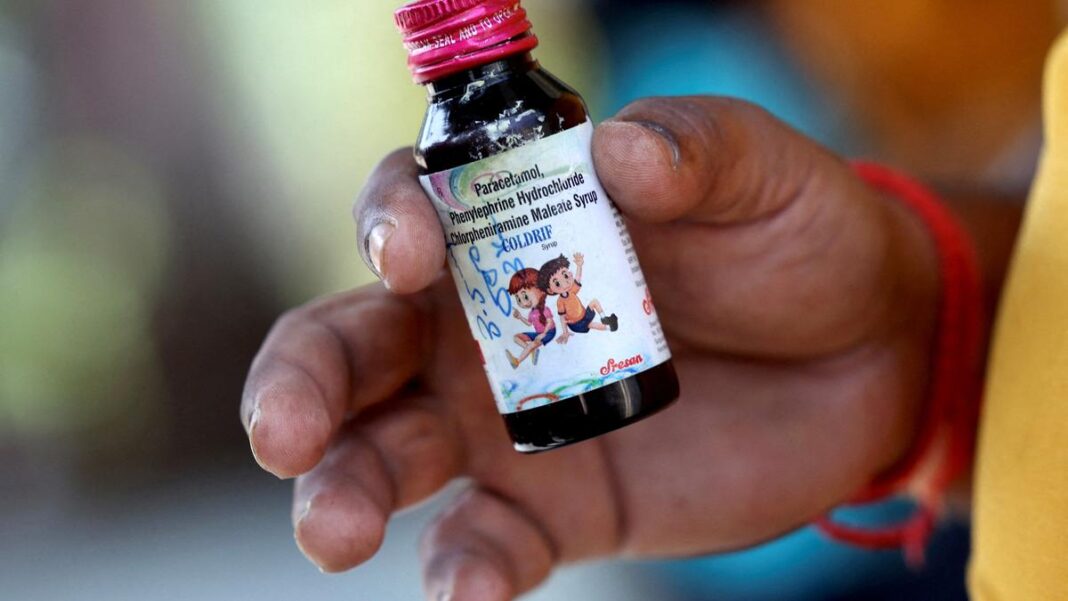
In a major development following the tragic deaths of several children in Madhya Pradesh, the owner of Sresan Pharma, a Chennai-based pharmaceutical company, has been arrested by police for allegedly manufacturing and distributing contaminated cough syrup.
According to reports, the cough syrup produced by Sresan Pharma was supplied to hospitals and local medical stores in Madhya Pradesh, where at least three children lost their lives after reportedly consuming the medicine. Preliminary investigations have suggested that the syrup contained high levels of toxic chemicals, making it unsafe for consumption.
Authorities in Madhya Pradesh had sent samples of the cough syrup for lab testing, and the results confirmed the presence of harmful substances, leading to an immediate ban on the product’s sale and distribution.
Following the lab confirmation, a special investigation team (SIT) tracked down the manufacturer in Chennai. The police then arrested the company’s owner, who is currently being questioned to uncover how such a dangerous product reached the market.
Health officials have also sealed Sresan Pharma’s manufacturing unit, citing multiple violations of drug safety norms and production guidelines. The Central Drugs Standard Control Organization (CDSCO) has joined the investigation to verify whether the company had valid licenses and followed quality control procedures.
This case has once again raised serious concerns over the quality of pharmaceutical manufacturing in India — especially among small and medium-scale drug producers. Officials say more arrests could follow as the investigation expands to include distributors and local officials who may have overlooked irregularities.
The Madhya Pradesh government has ordered a statewide inspection of all syrup-based medicines being sold in the market. Families affected by the incident have demanded strict action against those responsible, calling for stronger checks to prevent such tragedies in the future.
Meanwhile, health experts have urged the public to avoid unverified or locally branded medicines and only purchase products approved by registered pharmacists.
The tragic deaths have not only shaken families but also reignited debate about India’s pharmaceutical regulation system, calling for immediate reforms to ensure that “Made in India” medicines remain safe and trustworthy both at home and abroad.